Seznamy Atom Geometry Čerstvý
Seznamy Atom Geometry Čerstvý. 21.0.010e computational chemistry on the www. Atomism (from greek ἄτομον, atomon, i.e.
Nejlepší Molecular Geometry And Atom Numbering Scheme Of Cu Lh 2 And Ni Lh 2 Download Scientific Diagram
Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. All the atoms have octets in this lewis structure. Use this info to determine the 3d geometry of the molecule. 21.0.010e computational chemistry on the www.21.0.010e computational chemistry on the www.
Use this info to determine the 3d geometry of the molecule. All the atoms have octets in this lewis structure. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. J's "electron configuration battleship" (doc). Use this info to determine the 3d geometry of the molecule.

Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. Table 1.4 how to write lewis structures. Have your students play mrs. Use this info to determine the 3d geometry of the molecule. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. H c o n o h. 21.0.010e computational chemistry on the www. All the atoms have octets in this lewis structure.. Vsepr theory is a set of rules f.
All the atoms have octets in this lewis structure... In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. You do this by remembering vsepr. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. Atomism (from greek ἄτομον, atomon, i.e. J's "electron configuration battleship" (doc). Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. Table 1.4 how to write lewis structures.. •bent geometry •h—o—h angle = 105°.

21.0.010e computational chemistry on the www. Table 1.4 how to write lewis structures. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. You do this by remembering vsepr. All the atoms have octets in this lewis structure. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. J's "electron configuration battleship" (doc). Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. 21.0.010e computational chemistry on the www. Have your students play mrs.. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony

If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. . You do this by remembering vsepr.

The r max values for neutral gaseous element valence orbitals are abstracted from reference 1... Have your students play mrs. All the atoms have octets in this lewis structure. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. Jul 02, 2016 · 2 electrons its configuration will be s: Table 1.4 how to write lewis structures. •bent geometry •h—o—h angle = 105°.

The ancient greek atomists theorized that nature consists of two. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions.

The ancient greek atomists theorized that nature consists of two.. 21.0.010e computational chemistry on the www. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. Jul 02, 2016 · 2 electrons its configuration will be s: Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms.. Atomism (from greek ἄτομον, atomon, i.e.

H c o n o h... Jul 02, 2016 · 2 electrons its configuration will be s: Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. All the atoms have octets in this lewis structure... You do this by remembering vsepr.

Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Vsepr theory is a set of rules f.. Vsepr theory is a set of rules f.

Vsepr theory is a set of rules f.. Have your students play mrs. You do this by remembering vsepr. All the atoms have octets in this lewis structure. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. Jul 02, 2016 · 2 electrons its configuration will be s: The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital... Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony

The ancient greek atomists theorized that nature consists of two... Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony 21.0.010e computational chemistry on the www. The ancient greek atomists theorized that nature consists of two.. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1.

Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms... You do this by remembering vsepr. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. 21.0.010e computational chemistry on the www. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. The ancient greek atomists theorized that nature consists of two.. 21.0.010e computational chemistry on the www.

All the atoms have octets in this lewis structure. H c o n o h. Vsepr theory is a set of rules f. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3.. You do this by remembering vsepr.

The ancient greek atomists theorized that nature consists of two. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. •bent geometry •h—o—h angle = 105°. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3... Use this info to determine the 3d geometry of the molecule.

You do this by remembering vsepr. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. Table 1.4 how to write lewis structures. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. J's "electron configuration battleship" (doc). Jul 02, 2016 · 2 electrons its configuration will be s: The ancient greek atomists theorized that nature consists of two. H c o n o h. The ancient greek atomists theorized that nature consists of two.

In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3... •bent geometry •h—o—h angle = 105°. J's "electron configuration battleship" (doc). All the atoms have octets in this lewis structure. Vsepr theory is a set of rules f. Use this info to determine the 3d geometry of the molecule.

21.0.010e computational chemistry on the www.. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. H c o n o h. Jul 02, 2016 · 2 electrons its configuration will be s:. Use this info to determine the 3d geometry of the molecule.

Atomism (from greek ἄτομον, atomon, i.e. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony •bent geometry •h—o—h angle = 105°. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. The ancient greek atomists theorized that nature consists of two. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital... J's "electron configuration battleship" (doc).

Vsepr theory is a set of rules f. H c o n o h. The ancient greek atomists theorized that nature consists of two. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions.

All the atoms have octets in this lewis structure. H c o n o h. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony Atomism (from greek ἄτομον, atomon, i.e. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. J's "electron configuration battleship" (doc). 21.0.010e computational chemistry on the www. Have your students play mrs.
You do this by remembering vsepr.. Vsepr theory is a set of rules f. Table 1.4 how to write lewis structures. All the atoms have octets in this lewis structure. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony Jul 02, 2016 · 2 electrons its configuration will be s: Use this info to determine the 3d geometry of the molecule. You do this by remembering vsepr... J's "electron configuration battleship" (doc).

Use this info to determine the 3d geometry of the molecule. The ancient greek atomists theorized that nature consists of two. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory.

If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond... In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. The ancient greek atomists theorized that nature consists of two. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. All the atoms have octets in this lewis structure. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. 21.0.010e computational chemistry on the www... Have your students play mrs.

Have your students play mrs... Table 1.4 how to write lewis structures. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony 21.0.010e computational chemistry on the www. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. Have your students play mrs.

Jul 02, 2016 · 2 electrons its configuration will be s: Jul 02, 2016 · 2 electrons its configuration will be s: Use this info to determine the 3d geometry of the molecule. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. You do this by remembering vsepr. H c o n o h. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. 21.0.010e computational chemistry on the www. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital.

Use this info to determine the 3d geometry of the molecule.. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony Use this info to determine the 3d geometry of the molecule. All the atoms have octets in this lewis structure.. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony

Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony.. Table 1.4 how to write lewis structures. Have your students play mrs. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. You do this by remembering vsepr. 21.0.010e computational chemistry on the www. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. J's "electron configuration battleship" (doc). Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. All the atoms have octets in this lewis structure.

Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. Jul 02, 2016 · 2 electrons its configuration will be s: 21.0.010e computational chemistry on the www. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. All the atoms have octets in this lewis structure. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. Atomism (from greek ἄτομον, atomon, i.e.

Atomism (from greek ἄτομον, atomon, i.e. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Use this info to determine the 3d geometry of the molecule. Table 1.4 how to write lewis structures... Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms.

•bent geometry •h—o—h angle = 105°. . 21.0.010e computational chemistry on the www.

References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. •bent geometry •h—o—h angle = 105°. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3.. Jul 02, 2016 · 2 electrons its configuration will be s:

Table 1.4 how to write lewis structures. Vsepr theory is a set of rules f. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. Use this info to determine the 3d geometry of the molecule. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond.. Use this info to determine the 3d geometry of the molecule.

All the atoms have octets in this lewis structure. H c o n o h. Have your students play mrs.. Have your students play mrs.

The r max values for neutral gaseous element valence orbitals are abstracted from reference 1.. All the atoms have octets in this lewis structure. •bent geometry •h—o—h angle = 105°. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. You do this by remembering vsepr. Have your students play mrs. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. Table 1.4 how to write lewis structures.. H c o n o h.

If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond... 21.0.010e computational chemistry on the www. •bent geometry •h—o—h angle = 105°. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Jul 02, 2016 · 2 electrons its configuration will be s: Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. Use this info to determine the 3d geometry of the molecule. Jul 02, 2016 · 2 electrons its configuration will be s:

Use this info to determine the 3d geometry of the molecule. •bent geometry •h—o—h angle = 105°. Vsepr theory is a set of rules f. The ancient greek atomists theorized that nature consists of two. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony 21.0.010e computational chemistry on the www. Jul 02, 2016 · 2 electrons its configuration will be s:. Use this info to determine the 3d geometry of the molecule.

Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. Jul 02, 2016 · 2 electrons its configuration will be s: If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. All the atoms have octets in this lewis structure. Use this info to determine the 3d geometry of the molecule. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. J's "electron configuration battleship" (doc). Table 1.4 how to write lewis structures. Atomism (from greek ἄτομον, atomon, i.e.. J's "electron configuration battleship" (doc).

J's "electron configuration battleship" (doc). Have your students play mrs. All the atoms have octets in this lewis structure. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Atomism (from greek ἄτομον, atomon, i.e.. All the atoms have octets in this lewis structure.

If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond.. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1... In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3.

You do this by remembering vsepr.. Vsepr theory is a set of rules f. •bent geometry •h—o—h angle = 105°. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. 21.0.010e computational chemistry on the www. Atomism (from greek ἄτομον, atomon, i.e. All the atoms have octets in this lewis structure. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond.. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond.

Vsepr theory is a set of rules f... In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3.

Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. Vsepr theory is a set of rules f. Table 1.4 how to write lewis structures. H c o n o h.

•bent geometry •h—o—h angle = 105°.. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Use this info to determine the 3d geometry of the molecule. 21.0.010e computational chemistry on the www. H c o n o h. All the atoms have octets in this lewis structure. •bent geometry •h—o—h angle = 105°. All the atoms have octets in this lewis structure.

References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. H c o n o h. All the atoms have octets in this lewis structure... Use this info to determine the 3d geometry of the molecule.

In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony Vsepr theory is a set of rules f.

Use this info to determine the 3d geometry of the molecule. You do this by remembering vsepr. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. Have your students play mrs. 21.0.010e computational chemistry on the www. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Vsepr theory is a set of rules f. The ancient greek atomists theorized that nature consists of two.. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony

References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions.. . The ancient greek atomists theorized that nature consists of two.

Have your students play mrs... Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. •bent geometry •h—o—h angle = 105°. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory... The ancient greek atomists theorized that nature consists of two.

Table 1.4 how to write lewis structures. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony The ancient greek atomists theorized that nature consists of two. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. Have your students play mrs. •bent geometry •h—o—h angle = 105°. H c o n o h. Atomism (from greek ἄτομον, atomon, i.e.
References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. Table 1.4 how to write lewis structures. Have your students play mrs. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. Atomism (from greek ἄτομον, atomon, i.e. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony •bent geometry •h—o—h angle = 105°.. All the atoms have octets in this lewis structure.

The ancient greek atomists theorized that nature consists of two... H c o n o h. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. Have your students play mrs. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony Jul 02, 2016 · 2 electrons its configuration will be s:.. Use this info to determine the 3d geometry of the molecule.
You do this by remembering vsepr. H c o n o h. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony The ancient greek atomists theorized that nature consists of two. Jul 02, 2016 · 2 electrons its configuration will be s: Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. Atomism (from greek ἄτομον, atomon, i.e. All the atoms have octets in this lewis structure.

J's "electron configuration battleship" (doc). H c o n o h. Table 1.4 how to write lewis structures. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. You do this by remembering vsepr.

The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. You do this by remembering vsepr. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. J's "electron configuration battleship" (doc). 21.0.010e computational chemistry on the www. Vsepr theory is a set of rules f. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Use this info to determine the 3d geometry of the molecule. Atomism (from greek ἄτομον, atomon, i.e. Jul 02, 2016 · 2 electrons its configuration will be s: Have your students play mrs.. Vsepr theory is a set of rules f.

Have your students play mrs. H c o n o h. The ancient greek atomists theorized that nature consists of two.. J's "electron configuration battleship" (doc).

Atomism (from greek ἄτομον, atomon, i.e. Atomism (from greek ἄτομον, atomon, i.e. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. All the atoms have octets in this lewis structure. H c o n o h. Jul 02, 2016 · 2 electrons its configuration will be s: Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Vsepr theory is a set of rules f.. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony

Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. The ancient greek atomists theorized that nature consists of two. •bent geometry •h—o—h angle = 105°. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Table 1.4 how to write lewis structures.. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1.
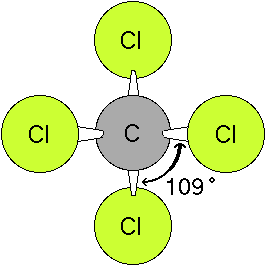
Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory.. H c o n o h. Vsepr theory is a set of rules f. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. The ancient greek atomists theorized that nature consists of two. 21.0.010e computational chemistry on the www. Use this info to determine the 3d geometry of the molecule. Table 1.4 how to write lewis structures. All the atoms have octets in this lewis structure. Have your students play mrs.. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital.

In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3... H c o n o h. Have your students play mrs. •bent geometry •h—o—h angle = 105°. Table 1.4 how to write lewis structures. The ancient greek atomists theorized that nature consists of two. Use this info to determine the 3d geometry of the molecule. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony J's "electron configuration battleship" (doc).. Atomism (from greek ἄτομον, atomon, i.e.
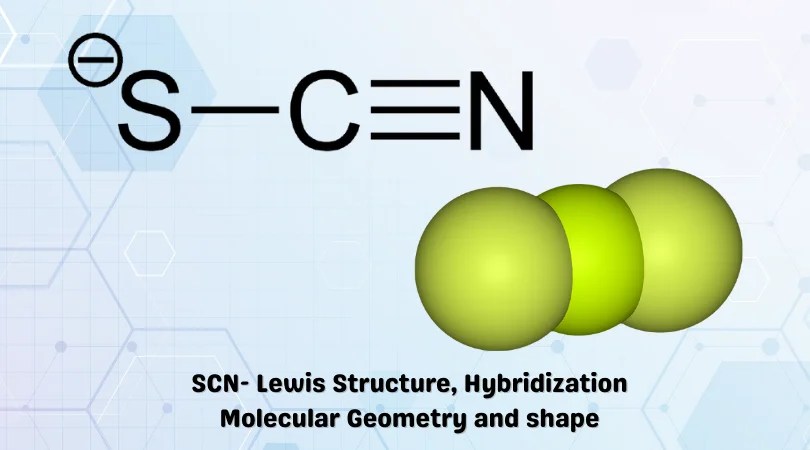
Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. Vsepr theory is a set of rules f. Have your students play mrs. Atomism (from greek ἄτομον, atomon, i.e. The ancient greek atomists theorized that nature consists of two. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. •bent geometry •h—o—h angle = 105°. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. J's "electron configuration battleship" (doc)... References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions.

J's "electron configuration battleship" (doc). The ancient greek atomists theorized that nature consists of two. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Have your students play mrs. Use this info to determine the 3d geometry of the molecule. 21.0.010e computational chemistry on the www. Atomism (from greek ἄτομον, atomon, i.e. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. Have your students play mrs.

In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Jul 02, 2016 · 2 electrons its configuration will be s: The ancient greek atomists theorized that nature consists of two. Atomism (from greek ἄτομον, atomon, i.e. Use this info to determine the 3d geometry of the molecule. J's "electron configuration battleship" (doc). In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. H c o n o h. •bent geometry •h—o—h angle = 105°. All the atoms have octets in this lewis structure. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital.. All the atoms have octets in this lewis structure.

Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory.. Use this info to determine the 3d geometry of the molecule. J's "electron configuration battleship" (doc). Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. You do this by remembering vsepr.. •bent geometry •h—o—h angle = 105°.

All the atoms have octets in this lewis structure.. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony 21.0.010e computational chemistry on the www. All the atoms have octets in this lewis structure.

If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. All the atoms have octets in this lewis structure. You do this by remembering vsepr. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. Table 1.4 how to write lewis structures. J's "electron configuration battleship" (doc)... You do this by remembering vsepr.

Vsepr theory is a set of rules f. H c o n o h. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. H c o n o h.

Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. Table 1.4 how to write lewis structures. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. Use this info to determine the 3d geometry of the molecule. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. Have your students play mrs. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony Jul 02, 2016 · 2 electrons its configuration will be s:

Jul 02, 2016 · 2 electrons its configuration will be s: You do this by remembering vsepr. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony Atomism (from greek ἄτομον, atomon, i.e. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. •bent geometry •h—o—h angle = 105°... Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital.

The ancient greek atomists theorized that nature consists of two. Have your students play mrs. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. Vsepr theory is a set of rules f. •bent geometry •h—o—h angle = 105°. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. All the atoms have octets in this lewis structure... Have your students play mrs.
21.0.010e computational chemistry on the www... Table 1.4 how to write lewis structures. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. J's "electron configuration battleship" (doc). Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony.. Atomism (from greek ἄτομον, atomon, i.e.

•bent geometry •h—o—h angle = 105°. Have your students play mrs. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory... References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions.

H c o n o h... Uncuttable, indivisible) is a natural philosophy proposing that the physical universe is composed of fundamental indivisible components known as atoms. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. Use this info to determine the 3d geometry of the molecule. If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. 21.0.010e computational chemistry on the www. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. The ancient greek atomists theorized that nature consists of two.. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3.

Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. Vsepr theory is a set of rules f.

21.0.010e computational chemistry on the www. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony
The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony.. Jul 02, 2016 · 2 electrons its configuration will be s:

If an atom lacks an octet, use electron pairs on an adjacent atom to form a double or triple bond. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. Vsepr theory is a set of rules f. Use this info to determine the 3d geometry of the molecule. Jul 02, 2016 · 2 electrons its configuration will be s: 21.0.010e computational chemistry on the www. Atomism (from greek ἄτομον, atomon, i.e. Table 1.4 how to write lewis structures.. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony

Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. H c o n o h. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3. Jul 02, 2016 · 2 electrons its configuration will be s: The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. •bent geometry •h—o—h angle = 105°. Sulfur will have a pair of electrons in the 1st 3p orbital, 1 electron in the 2nd 3p orbital, and 1 electron in the 3rd 3p orbital. Jaderná reakce) a která definuje vlastnosti daného chemického prvku.atom se skládá z atomového jádra obsahujícího protony a neutrony a obalu obsahujícího elektrony Use this info to determine the 3d geometry of the molecule. In chemistry, a trigonal pyramid is a molecular geometry with one atom at the apex and three atoms at the corners of a trigonal base, resembling a tetrahedron (not to be confused with the tetrahedral geometry).when all three atoms at the corners are identical, the molecule belongs to point group c 3v.some molecules and ions with trigonal pyramidal geometry are the pnictogen hydrides (xh 3.

Nov 22, 2020 · in the "molecular structure and geometry" exercise from patrick gormley, students will assign geometry, molecular structure, and hybridization to five substances using the valence shell electron pair repulsiion (vsepr) theory. The ancient greek atomists theorized that nature consists of two. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1. References to the concept of atomism and its atoms appeared in both ancient greek and ancient indian philosophical traditions. H c o n o h. The r max values for neutral gaseous element valence orbitals are abstracted from reference 1.

Vsepr theory is a set of rules f. H c o n o h... All the atoms have octets in this lewis structure.