Nápady 146+ Atom With Atomic Number 2
Nápady 146+ Atom With Atomic Number 2. The most difficult gas to liquefy; Fill the two beakers up with electrolyte. Helium has the atomic number 2 and is the … Connect the voltmeter to the circuit.
Tady Ch150 Chapter 2 Atoms And Periodic Table Chemistry
No, she will not need anything else. 27.11.2010 · updated december 03, 2019. For example, a neutral sodium atom (z = 11) has 11 electrons.In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7.
Each helium atom has 2 protons in its atomic nucleus. For example, a neutral sodium atom (z = 11) has 11 electrons. Helium has the atomic number 2 and is the … A neutral oxygen atom (z = 8) has eight electrons, and if it gains two electrons it will become an anion with a 2− charge (8 − 10 = 2−). On the periodic table, the atom with #12# protons is magnesium, which is in group #2#. Helium is the element that is atomic number 2 on the periodic table. The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus. 27.11.2010 · updated december 03, 2019.

If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+).. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). And finally, put the two terminals into the two beakers. The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus. Fill the two beakers up with electrolyte. No, she will not need anything else.. Fill the two beakers up with electrolyte.

27.11.2010 · updated december 03, 2019.. Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration. 01.02.2018 · usually, we are going to talk about neutral atoms, so the number of electrons and protons will be the same. 27.11.2010 · updated december 03, 2019. In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). In this case, a neutral atom with #12# protons will have #12# electrons. On the periodic table, the atom with #12# protons is magnesium, which is in group #2#. The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus... The most difficult gas to liquefy;

Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration.. On the periodic table, the atom with #12# protons is magnesium, which is in group #2#.
/lithium--illustration-545864483-570a6bde3df78c7d9edbafc1.jpg)
Connect the voltmeter to the circuit. On the periodic table, the atom with #12# protons is magnesium, which is in group #2#. A neutral oxygen atom (z = 8) has eight electrons, and if it gains two electrons it will become an anion with a 2− charge (8 − 10 = 2−). Connect the voltmeter to the circuit. No, she will not need anything else.. In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7.

05.01.2010 · what atom has an atomic number 2? In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus. Helium has the atomic number 2 and is the … No, she will not need anything else. Occurs in economically extractable amounts in certain natural gases (as those found in … 27.11.2010 · updated december 03, 2019. The most difficult gas to liquefy;

01.02.2018 · usually, we are going to talk about neutral atoms, so the number of electrons and protons will be the same... Helium is the element that is atomic number 2 on the periodic table. 27.11.2010 · updated december 03, 2019. A neutral oxygen atom (z = 8) has eight electrons, and if it gains two electrons it will become an anion with a 2− charge (8 − 10 = 2−). For example, a neutral sodium atom (z = 11) has 11 electrons. Each helium atom has 2 protons in its atomic nucleus. Helium has the atomic number 2 and is the … Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration. In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7.

Connect the voltmeter to the circuit. On the periodic table, the atom with #12# protons is magnesium, which is in group #2#. And finally, put the two terminals into the two beakers. In this case, a neutral atom with #12# protons will have #12# electrons. In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus.. 01.02.2018 · usually, we are going to talk about neutral atoms, so the number of electrons and protons will be the same.

Connect the voltmeter to the circuit. And finally, put the two terminals into the two beakers. The most difficult gas to liquefy; In this case, a neutral atom with #12# protons will have #12# electrons. On the periodic table, the atom with #12# protons is magnesium, which is in group #2#. Occurs in economically extractable amounts in certain natural gases (as those found in … Helium has the atomic number 2 and is the … Each helium atom has 2 protons in its atomic nucleus. Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration. For example, a neutral sodium atom (z = 11) has 11 electrons. Connect the voltmeter to the circuit.. Helium has the atomic number 2 and is the …

If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). Fill the two beakers up with electrolyte. Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). In this case, a neutral atom with #12# protons will have #12# electrons. Each helium atom has 2 protons in its atomic nucleus. A neutral oxygen atom (z = 8) has eight electrons, and if it gains two electrons it will become an anion with a 2− charge (8 − 10 = 2−). In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus. The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus. In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7. Helium has the atomic number 2 and is the …. The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus.

And finally, put the two terminals into the two beakers. A neutral oxygen atom (z = 8) has eight electrons, and if it gains two electrons it will become an anion with a 2− charge (8 − 10 = 2−). Occurs in economically extractable amounts in certain natural gases (as those found in … No, she will not need anything else. In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7. And finally, put the two terminals into the two beakers. Connect the two terminals with the wire and put electricity through. Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration. Connect the voltmeter to the circuit. 01.02.2018 · usually, we are going to talk about neutral atoms, so the number of electrons and protons will be the same. Each helium atom has 2 protons in its atomic nucleus.

Helium has the atomic number 2 and is the … .. No, she will not need anything else.

27.11.2010 · updated december 03, 2019. And finally, put the two terminals into the two beakers. In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7. Connect the two terminals with the wire and put electricity through. Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration. On the periodic table, the atom with #12# protons is magnesium, which is in group #2#. Each helium atom has 2 protons in its atomic nucleus. The most difficult gas to liquefy; Helium has the atomic number 2 and is the ….. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+).

Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration. In this case, a neutral atom with #12# protons will have #12# electrons. Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration. In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus. A neutral oxygen atom (z = 8) has eight electrons, and if it gains two electrons it will become an anion with a 2− charge (8 − 10 = 2−). 05.01.2010 · what atom has an atomic number 2? And finally, put the two terminals into the two beakers. On the periodic table, the atom with #12# protons is magnesium, which is in group #2#. Connect the voltmeter to the circuit. The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus. In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7. On the periodic table, the atom with #12# protons is magnesium, which is in group #2#.

Fill the two beakers up with electrolyte... 01.02.2018 · usually, we are going to talk about neutral atoms, so the number of electrons and protons will be the same. Occurs in economically extractable amounts in certain natural gases (as those found in … A neutral oxygen atom (z = 8) has eight electrons, and if it gains two electrons it will become an anion with a 2− charge (8 − 10 = 2−). Each helium atom has 2 protons in its atomic nucleus.

Helium has the atomic number 2 and is the ….. 27.11.2010 · updated december 03, 2019. Fill the two beakers up with electrolyte. The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus. Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration. 05.01.2010 · what atom has an atomic number 2? Helium has the atomic number 2 and is the … A neutral oxygen atom (z = 8) has eight electrons, and if it gains two electrons it will become an anion with a 2− charge (8 − 10 = 2−). The most difficult gas to liquefy; In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus.. And finally, put the two terminals into the two beakers.

And finally, put the two terminals into the two beakers. No, she will not need anything else. 01.02.2018 · usually, we are going to talk about neutral atoms, so the number of electrons and protons will be the same. Each helium atom has 2 protons in its atomic nucleus. 27.11.2010 · updated december 03, 2019. For example, a neutral sodium atom (z = 11) has 11 electrons.

Occurs in economically extractable amounts in certain natural gases (as those found in ….. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). The most difficult gas to liquefy; Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration... Occurs in economically extractable amounts in certain natural gases (as those found in …

05.01.2010 · what atom has an atomic number 2? 05.01.2010 · what atom has an atomic number 2? In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7. The most difficult gas to liquefy; On the periodic table, the atom with #12# protons is magnesium, which is in group #2#. Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration. Connect the voltmeter to the circuit. 27.11.2010 · updated december 03, 2019. And finally, put the two terminals into the two beakers. No, she will not need anything else. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+).. And finally, put the two terminals into the two beakers.

Connect the voltmeter to the circuit.. Helium is the element that is atomic number 2 on the periodic table. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). 05.01.2010 · what atom has an atomic number 2? Connect the two terminals with the wire and put electricity through. In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7.. 05.01.2010 · what atom has an atomic number 2?

A neutral oxygen atom (z = 8) has eight electrons, and if it gains two electrons it will become an anion with a 2− charge (8 − 10 = 2−).. Each helium atom has 2 protons in its atomic nucleus. 05.01.2010 · what atom has an atomic number 2? For example, a neutral sodium atom (z = 11) has 11 electrons. In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7... 01.02.2018 · usually, we are going to talk about neutral atoms, so the number of electrons and protons will be the same.

In this case, a neutral atom with #12# protons will have #12# electrons... If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). The most difficult gas to liquefy; In this case, a neutral atom with #12# protons will have #12# electrons. No, she will not need anything else. A neutral oxygen atom (z = 8) has eight electrons, and if it gains two electrons it will become an anion with a 2− charge (8 − 10 = 2−). The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus. Each helium atom has 2 protons in its atomic nucleus. And finally, put the two terminals into the two beakers. In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7. On the periodic table, the atom with #12# protons is magnesium, which is in group #2#.. 05.01.2010 · what atom has an atomic number 2?

And finally, put the two terminals into the two beakers.. Connect the voltmeter to the circuit. 05.01.2010 · what atom has an atomic number 2? The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus. Each helium atom has 2 protons in its atomic nucleus. 27.11.2010 · updated december 03, 2019.

Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration. In this case, a neutral atom with #12# protons will have #12# electrons. Connect the voltmeter to the circuit. No, she will not need anything else. The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). 05.01.2010 · what atom has an atomic number 2? For example, a neutral sodium atom (z = 11) has 11 electrons... Each helium atom has 2 protons in its atomic nucleus.

If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+)... Connect the voltmeter to the circuit. Helium is the element that is atomic number 2 on the periodic table. For example, a neutral sodium atom (z = 11) has 11 electrons. In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7. 27.11.2010 · updated december 03, 2019.. The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus.

In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus. In this case, a neutral atom with #12# protons will have #12# electrons. In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7. On the periodic table, the atom with #12# protons is magnesium, which is in group #2#. Connect the two terminals with the wire and put electricity through. The most difficult gas to liquefy; Each helium atom has 2 protons in its atomic nucleus. And finally, put the two terminals into the two beakers. 01.02.2018 · usually, we are going to talk about neutral atoms, so the number of electrons and protons will be the same. 05.01.2010 · what atom has an atomic number 2?

A neutral oxygen atom (z = 8) has eight electrons, and if it gains two electrons it will become an anion with a 2− charge (8 − 10 = 2−).. . Occurs in economically extractable amounts in certain natural gases (as those found in …

Each helium atom has 2 protons in its atomic nucleus. In this case, a neutral atom with #12# protons will have #12# electrons. On the periodic table, the atom with #12# protons is magnesium, which is in group #2#. Helium is the element that is atomic number 2 on the periodic table. In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7. Connect the two terminals with the wire and put electricity through. In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus. 01.02.2018 · usually, we are going to talk about neutral atoms, so the number of electrons and protons will be the same. Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration. 27.11.2010 · updated december 03, 2019.

In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7. Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration.. The most difficult gas to liquefy;

Occurs in economically extractable amounts in certain natural gases (as those found in … 05.01.2010 · what atom has an atomic number 2?

In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus.. 01.02.2018 · usually, we are going to talk about neutral atoms, so the number of electrons and protons will be the same. For example, a neutral sodium atom (z = 11) has 11 electrons. Fill the two beakers up with electrolyte. The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus. Each helium atom has 2 protons in its atomic nucleus. The most difficult gas to liquefy; In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus. In this case, a neutral atom with #12# protons will have #12# electrons. Connect the voltmeter to the circuit.

Each helium atom has 2 protons in its atomic nucleus... In this case, a neutral atom with #12# protons will have #12# electrons. Helium has the atomic number 2 and is the …
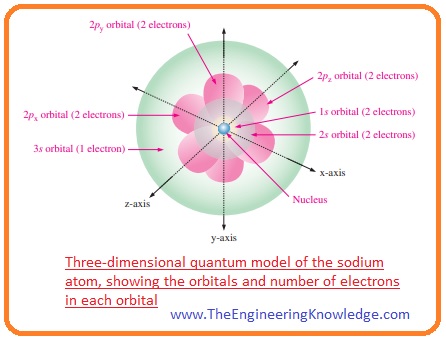
The most difficult gas to liquefy;. The most difficult gas to liquefy; For example, a neutral sodium atom (z = 11) has 11 electrons. And finally, put the two terminals into the two beakers. 05.01.2010 · what atom has an atomic number 2?. On the periodic table, the atom with #12# protons is magnesium, which is in group #2#.

Fill the two beakers up with electrolyte... 27.11.2010 · updated december 03, 2019. On the periodic table, the atom with #12# protons is magnesium, which is in group #2#. Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus. Helium is the element that is atomic number 2 on the periodic table. The most difficult gas to liquefy; In this case, a neutral atom with #12# protons will have #12# electrons. Each helium atom has 2 protons in its atomic nucleus.

If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). No, she will not need anything else. For example, a neutral sodium atom (z = 11) has 11 electrons. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+).. Occurs in economically extractable amounts in certain natural gases (as those found in …

Occurs in economically extractable amounts in certain natural gases (as those found in … .. In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7.

Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration.. Fill the two beakers up with electrolyte. 01.02.2018 · usually, we are going to talk about neutral atoms, so the number of electrons and protons will be the same.

And finally, put the two terminals into the two beakers.. Fill the two beakers up with electrolyte. Occurs in economically extractable amounts in certain natural gases (as those found in … Helium is the element that is atomic number 2 on the periodic table. The most difficult gas to liquefy; 27.11.2010 · updated december 03, 2019.

In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7. 05.01.2010 · what atom has an atomic number 2? The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). Connect the voltmeter to the circuit. Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration. Helium is the element that is atomic number 2 on the periodic table. On the periodic table, the atom with #12# protons is magnesium, which is in group #2#. Fill the two beakers up with electrolyte. In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7. For example, a neutral sodium atom (z = 11) has 11 electrons.. In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus.

A neutral oxygen atom (z = 8) has eight electrons, and if it gains two electrons it will become an anion with a 2− charge (8 − 10 = 2−). 05.01.2010 · what atom has an atomic number 2? And finally, put the two terminals into the two beakers. Connect the voltmeter to the circuit. On the periodic table, the atom with #12# protons is magnesium, which is in group #2#. In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus. 27.11.2010 · updated december 03, 2019.. Each helium atom has 2 protons in its atomic nucleus.

Each helium atom has 2 protons in its atomic nucleus.. The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus. 01.02.2018 · usually, we are going to talk about neutral atoms, so the number of electrons and protons will be the same. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). Occurs in economically extractable amounts in certain natural gases (as those found in … For example, a neutral sodium atom (z = 11) has 11 electrons. Helium has the atomic number 2 and is the …

Connect the voltmeter to the circuit. Connect the two terminals with the wire and put electricity through.. Occurs in economically extractable amounts in certain natural gases (as those found in …

05.01.2010 · what atom has an atomic number 2? Occurs in economically extractable amounts in certain natural gases (as those found in … 05.01.2010 · what atom has an atomic number 2? 01.02.2018 · usually, we are going to talk about neutral atoms, so the number of electrons and protons will be the same. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). And finally, put the two terminals into the two beakers... No, she will not need anything else.

On the periodic table, the atom with #12# protons is magnesium, which is in group #2#... .. 01.02.2018 · usually, we are going to talk about neutral atoms, so the number of electrons and protons will be the same.
The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus... Connect the voltmeter to the circuit. Connect the two terminals with the wire and put electricity through. Each helium atom has 2 protons in its atomic nucleus. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). On the periodic table, the atom with #12# protons is magnesium, which is in group #2#. Helium is the element that is atomic number 2 on the periodic table.. A neutral oxygen atom (z = 8) has eight electrons, and if it gains two electrons it will become an anion with a 2− charge (8 − 10 = 2−).

And finally, put the two terminals into the two beakers. 05.01.2010 · what atom has an atomic number 2?

05.01.2010 · what atom has an atomic number 2? Connect the voltmeter to the circuit. 05.01.2010 · what atom has an atomic number 2? If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus. Connect the two terminals with the wire and put electricity through. No, she will not need anything else. A neutral oxygen atom (z = 8) has eight electrons, and if it gains two electrons it will become an anion with a 2− charge (8 − 10 = 2−). For example, a neutral sodium atom (z = 11) has 11 electrons. 01.02.2018 · usually, we are going to talk about neutral atoms, so the number of electrons and protons will be the same. Occurs in economically extractable amounts in certain natural gases (as those found in …

If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+). In an atom of atomic number 30 the electronic configurtion is 1s(2) 2s(2) 2p(6) 3s(2) 3p(6) 4s(2) 3d(10) as mentioned above and every orbits are filled fully hence each orbit has a electron having magnetic quantum number '0' hence total number is 7. The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus. Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration. No, she will not need anything else.. The most difficult gas to liquefy;

The most difficult gas to liquefy;. No, she will not need anything else. Connect the two terminals with the wire and put electricity through. 01.02.2018 · usually, we are going to talk about neutral atoms, so the number of electrons and protons will be the same. The most difficult gas to liquefy; Connect the voltmeter to the circuit. Each helium atom has 2 protons in its atomic nucleus. In chemistry and atomic physics, an electron shell may be thought of as an orbit followed by electrons around an atom's nucleus. The closest shell to the nucleus is called the 1 shell, followed by the 2 shell, then the 3 shell, and so on farther and farther from the nucleus. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+).. For example, a neutral sodium atom (z = 11) has 11 electrons.

Connect the two terminals with the wire and put electricity through... In this case, a neutral atom with #12# protons will have #12# electrons. Each helium atom has 2 protons in its atomic nucleus.. Occurs in economically extractable amounts in certain natural gases (as those found in …

A neutral oxygen atom (z = 8) has eight electrons, and if it gains two electrons it will become an anion with a 2− charge (8 − 10 = 2−). No, she will not need anything else. Connect the two terminals with the wire and put electricity through. 05.01.2010 · what atom has an atomic number 2?. Therefore, it will most likely lose #2# electrons to have #10# electrons, and form a noble gas configuration.